What is PIFELTRO?
PIFELTRO is a prescription HIV-1 medicine used with other antiretroviral medicines to treat human immunodeficiency virus-1 (HIV-1) infection in adults.
HIV-1 is the virus that causes AIDS (Acquired Immune Deficiency Syndrome).
PIFELTRO may be right for you if you have not received HIV-1 medicines in the past or if your healthcare provider determines that you meet certain requirements to replace your current HIV-1 medicines.
Learn about:
Side effects
Managing your
HIV-1
Cholesterol and
PIFELTRO
A low rate of side effects
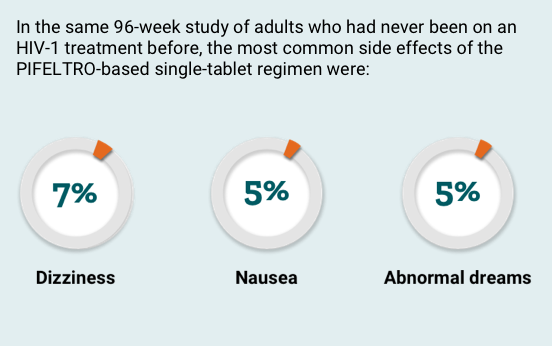
In a 96-week study of adults who had never been on an HIV-1 treatment before, 364 adults received a PIFELTRO-based single-tablet regimen, while 364 adults received an Atripla once-daily tablet. In this study, fewer adults stopped taking a PIFELTRO-based single-tablet regimen than Atripla because of side effects.
What are the most common side effects?
The most common side effects of PIFELTRO include:
- nausea
- dizziness
- headache
- tiredness
- diarrhea
- stomach (abdominal) pain
- abnormal dreams
These are not all the possible side effects of PIFELTRO. Call your healthcare provider for medical advice about side effects. You may report side effects to the FDA at 1-800-FDA-1088.
Changes in your immune system (immune reconstitution syndrome) can happen when you start taking HIV-1 medicines. Your immune system may get stronger and begin to fight infections that have been hidden in your body for a long time. Tell your healthcare provider right away if you start having new symptoms after starting your HIV-1 medicine.
Your health and HIV-1
Staying undetectable
PIFELTRO, when taken as part of a multi-tablet regimen, can help you get your viral load to undetectable.
Undetectable means the level of HIV-1 in your blood is so low that lab tests can’t detect it.
And when your HIV-1 is under control, you can focus on your health and life goals beyond HIV-1.

In a 48-week study, adults living with HIV-1 who were undetectable on their current treatment regimen switched to a PIFELTRO-based single-tablet regimen either at the start of the study, or at week 24 of the study.
At the start of the study:
- 447 adults switched to a PIFELTRO-based single-tablet regimen.
- 91% of those adults stayed undetectable at week 48.
At week 24 of the study:
- 209 adults switched to a PIFELTRO-based single-tablet regimen.
- 95% of those adults were undetectable at week 24 before switching to a PIFELTRO-based single-tablet regimen.
At week 48 of the study:
- 95% of those adults who switched to PIFELTRO-based single-tablet regimen at week 24 stayed undetectable at week 48.
PIFELTRO may not have these effects on all adults living with HIV-1.
More about this study:
Patients were undetectable (HIV-1 RNA <50 copies/mL) for at least 6 months before starting the trial and had no history of resistancea to the PIFELTRO-based single-tablet regimen. The current treatment regimens consisted of 2 nucleoside reverse transcriptase inhibitors (NRTIs) in combination with a protease inhibitor (PI) plus either ritonavir or cobicistat, elvitegravir plus cobicistat, or a non-nucleoside reverse transcriptase inhibitor (NNRTI) before switching to a PIFELTRO-based single-tablet regimen. Patients were assigned by chance to one of the two treatment groups in the study. Patients and their healthcare providers taking part in the study knew which group they were part of.
aResistance happens when the virus mutates and your medicine stops working.
About cholesterol
HIV-1 and/or treatments for HIV may have a negative impact on your “bad” cholesterol (LDL-C). In a 96-week study of adults who had never been on an HIV-1 treatment before, 320 adults received PIFELTRO as part of a treatment regimen and 311 adults received another treatment regimen.b
PIFELTRO, as part of a treatment regimen, did not raise “bad” cholesterol, while the other treatment regimen did.
The benefit of these changes has not been demonstrated.
Effect on “bad” cholesterol (LDL-C) at week 48:
bThe other treatment regimen included darunavir plus a ritonavir boost (PI + ritonavir boost), once daily.